Alvotech: Biosimilar Specialist Targeting Lucrative Markets (NASDAQ:ALVO)
Extreme Media/E+ via Getty Images
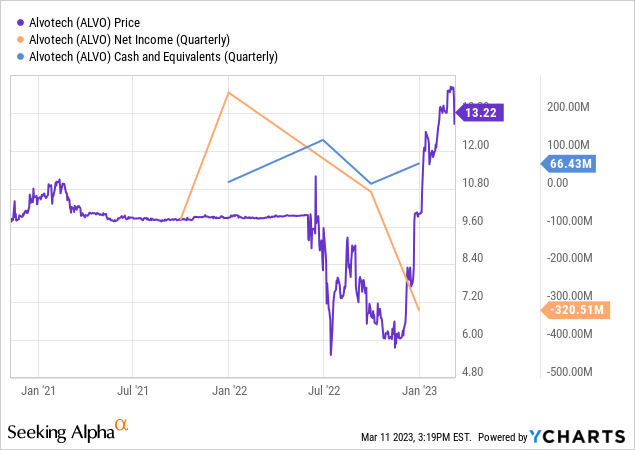
Alvotech’s (NASDAQ:ALVO) launch of its first biosimilar product, a Humira (adalimumab) equivalent, in Europe and Canada, along with the pending approval for the United States, has established Alvotech as a leader in the biosimilar industry. The company has reported impressive financial figures that support its strategy execution — $85 million in sales in FY 2022, and an enviable cash position of $66.4 million.
Despite potential hurdles ahead such as high development costs or competing manufacturers, Alvotech is well-positioned due to its end-to-end platform which allows it to work on multiple biosimilars at once. When combined with a solid base of finances, strategic partnerships, and a promising pipeline outlook, Alvotech emerges as an attractive investment option for those looking to enter the biosimilar market.
The Financial Landscape
Despite reporting a net loss of $513.6 million for the year ended December 31, 2022, Alvotech has made significant progress in developing its biosimilars, launching its first product, AVT02, and securing multiple licensing agreements. These positive developments have set the stage for a successful future for Alvotech, making it an attractive investment for investors who are willing to look beyond the current losses and recognize the company’s potential.
ycharts.com
One of the key factors contributing to Alvotech’s potential success is its strong cash position. The company had cash and cash equivalents of $66.4 million by the end of FY 22, which gives it a solid foundation for future growth. In addition, Alvotech has borrowings of $764.6 million, including $19.9 million in current borrowings, as of the end of 2022. This gives the company the necessary liquidity to fund its ongoing R&D activities and commercial launches, as well as any potential future acquisitions or partnerships.
Alvotech has also reported strong revenue growth, with revenue, including other income, totaling $85.0 million for FY 22, compared to $39.7 million for the full year 2021. The increase in revenue was primarily driven by the successful launch of AVT02 in several countries in Europe and Canada, as well as licensing revenue for AVT05 and AVT04. With the company’s strong pipeline of biosimilar candidates and licensing agreements, Alvotech is well-positioned for continued revenue growth in the coming years.
While the company’s cost of product revenue was disproportionately high relative to its product revenue, Alvotech expects to stabilize due to scaling and expansion of new launches. As the company continues to grow and expand its product portfolio, it is reasonable to expect that its costs will decrease as production increases. In addition, the company’s R&D expenses have decreased, primarily due to the recognition of manufacturing costs as cost of product revenue following the first commercial launch of AVT02.
AVT05 Pharmacokinetic Study
Alvotech has announced a pharmacokinetic study for AVT05. It is a biosimilar candidate to Simponi and Simponi Aria (golimumab), which is used to treat autoimmune diseases. In addition to pharmacokinetics, the safety, and tolerability of AVT05 in comparison to Simponi in healthy adult subjects will also be studied. The progress of the AVT05 development program, which marks the entry of the company’s next biosimilar candidate into clinical studies should be seen as a positive sign for investors.
The market potential for AVT05 is significant, with global sales by Simponi and Simponi Aria exceeding $2.2 billion in the past year. AVT05 is an investigational product that has not yet been approved in any country. It works by blocking tumor necrosis factor-alpha (TNF alpha), a protein involved in diseases such as rheumatoid and psoriatic arthritis, and ankylosing spondylitis. Despite its current status, the potential for regulatory approval of AVT05 is high due to Alvotech’s success in developing biosimilar candidates.
Fuji Pharma Partnership
Alvotech and Fuji Pharma, a Japanese pharmaceutical company, have decided to expand their exclusive commercialization partnership to include a new undisclosed biosimilar candidate. The existing partnership includes the new candidate as well as seven other biosimilars that are then commercialized in Japan by Fuji. The extension of this partnership reflects the common objective of expanding the accessibility of biosimilars to Japan’s market.
The Japanese market potential is significant, and the expansion of the partnership between Alvotech and Fuji provides an opportunity for significant growth in this market. Alvotech’s expansion of its partnership with Fuji is a positive development for the company and investors.
EMA Acceptance of AVT04 Authorization
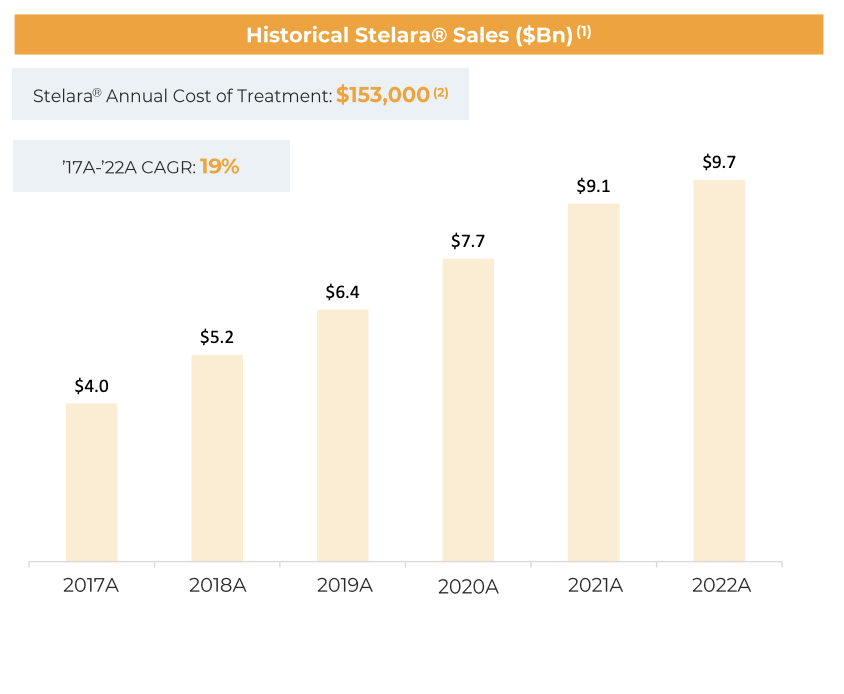
Alvotech and STADA Arzneimittel, a worldwide pharmaceutical company, have declared that AVT04, which is Alvotech’s suggested biosimilar for Stelara (ustekinumab), has had its Marketing Authorization Application accepted by the European Medicines Agency (EMA). The two companies hope that the EMA could endorse granting a marketing authorization for AVT04 in the second half of 2023.
investors.alvotech.com/news-events/events
If ustekinumab receives authorization, it will expand the already broad collection of six endorsed biosimilars that STADA has available in Europe. This range comprises a citrate-free adalimumab with a high concentration, which the company brought to market through its cooperation with Alvotech.
In 2019, Alvotech and STADA made an announcement about their strategic partnership to market eight biosimilar candidates created by Alvotech in Europe. By December 2022, the two companies had already introduced the initial biosimilar in their partnership, high-concentration adalimumab, for marketing and sales in 16 countries in Europe.
The EMA’s acceptance of Alvotech’s Marketing Authorization Application for AVT04 is a positive development for the company and investors. The approval of AVT04 will expand Alvotech’s product portfolio and increase patient access to affordable biologic medicines.
Competition Among Biosimilar Companies
While there are many other biotech companies that operate in this space, one of the companies that Alvotech is superior to is Coherus BioSciences (CHRS).
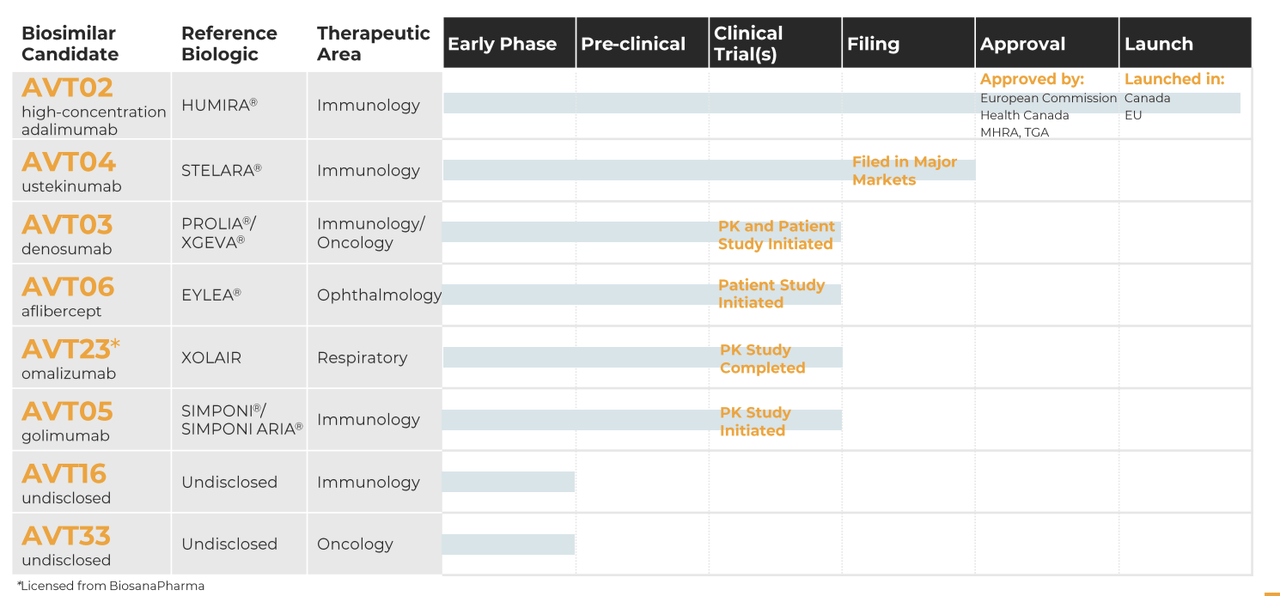
One key area where Alvotech excels over Coherus Biosciences is its strong pipeline of biosimilar candidates. Alvotech has a portfolio of eight products and product candidates, with three biosimilar candidates entering clinical development in 2022. This indicates that Alvotech is continually investing in research and development, which is crucial for maintaining a strong pipeline of products.
investors.alvotech.com/news-events/events
In contrast, Coherus Biosciences has a portfolio of three biosimilar products, which is relatively small compared to Alvotech. Another area where Alvotech excels is its focus on expanding patient access to affordable biologic medicines. The company’s end-to-end biosimilars platform is specifically designed to enable the simultaneous development and manufacture of multiple products, from the beginning to the end of the process. By doing so, Alvotech can offer high-quality biosimilar medicines at a lower cost, making it easier for patients to access these treatments.
Finally, Alvotech’s strong partnerships with global pharmaceutical companies, such as STADA Arzneimittel and Fuji Pharma, are a key advantage. These partnerships provide Alvotech with access to new markets and distribution channels, as well as the resources and expertise of established pharmaceutical companies.
Potential Limitations and Pitfalls
One potential counterargument to the bullish view of Alvotech’s products is the fact that biosimilars face a higher regulatory burden than traditional small-molecule drugs. Biosimilars are required to demonstrate their similarity to the reference product in terms of safety, efficacy, and quality, which can be a lengthy and costly process. This can lead to delays in the approval and commercialization of biosimilars, as well as increased costs for development and manufacturing.
Another potential issue is the threat of litigation from originator companies. Originator companies have been known to file patent infringement lawsuits in an attempt to delay the launch of biosimilars, which can further delay the availability of affordable treatments for patients.
To address these counterarguments, Alvotech has taken a strategic approach to its product development and commercialization. The company has a robust end-to-end biosimilar platform that is designed to support the efficient and cost-effective development and manufacturing of multiple products simultaneously. This approach enables Alvotech to potentially reduce costs associated with development and manufacturing and to bring biosimilars to market faster than competitors.
Additionally, Alvotech has a strong intellectual property strategy and has filed multiple patent applications related to its biosimilar products. This strategy can potentially protect the company from patent infringement lawsuits from originator companies.
Key Takeaways
Alvotech’s financial outlook appears promising with its strong cash position, growing revenue, and expanding product portfolio. The company’s strategic partnerships and collaborations with established pharmaceutical companies also indicate a bright future. Despite potential challenges, such as regulatory hurdles and competition, Alvotech’s innovative approach to developing and manufacturing biosimilars positions it for success in the ever-growing biotech industry. As more and more patients and healthcare providers demand affordable, high-quality biologics, Alvotech is well-positioned to meet this need with its cost-effective biosimilar products.
No Byline Policy
Editorial Guidelines
Corrections Policy
Source
